
Rouging When Stainless Steel Corrodes
Excerpt form the GMP Compliance Adviser, Chapter 5.E Operation and maintenance of water systems
12 min. reading time | by Herbert Bendlin
Published in LOGFILE 10/2021
What is rouging?
Rouging is a phenomenon that frequently occurs in water systems constructed from austenitic CrNi steel. The causes of rouging have been largely clarified. The mechanism of rouging formation and the parameters that have an influence have been described in the literature. The ISPE Newsletter No.1 (2007)1 and a number of different expert articles (see 5.H References) contain detailed descriptions of the phenomenon. It has been shown that a ferrite content up to 3% does not have a significant impact on the occurrence of rouging.
The GMP regulations do not cover rouging. The ISPE Baseline Water and Steam contains a small amount of information which is not very helpful. It recommends reducing the temperature and protecting the system through regular cleaning and repassivation. The latter has proven successful in practice.
In order to understand the phenomenon of rouging, knowledge about the surface of stainless steel is helpful. Stainless steel surfaces that do not show signs of corrosion are always shiny and with no sign of deposits. A transparent passive film which is 1–3 nm thick and contains a large amount of chromium oxide provides sufficient and reliable protection from corrosive interactions.
Figure 5.E-3 contains a schematic representation of the chemical and physical interactions. Figure 5.E-4 shows the protective effect of the film and the damage caused by corrosion.
Figure 5.E-3 Structure of the passive film on stainless steel alloys
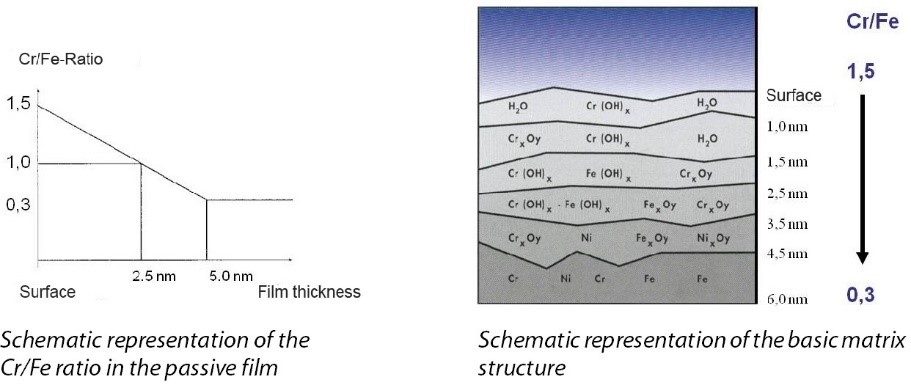
Figure 5.E-4 Schematic representation of a metal surface with intact passive film (left) and with a partially damaged passive film and (the start of) corrosion (right) (Source: Henkel Beiz- und Elektropoliertechnik GmbH & Co.KG, 19306 Neustadt-Glewe, Germany)

The positive properties of an intact passive film include:
- prevention of corrosion under normal conditions
- quick and safe surface restoration (repassivation) if sufficient chrome and oxygen is available
- impermeable for ions (e.g. chloride ions)
If the passive film is damaged or removed, normal oxidation occurs on the iron-chrome alloy. This is immediately recognisable as a reddish brown particulate precipitate on surfaces in hot ultrapure water systems (Figure 5.E-5).
This type of rouge coating is easily wiped off. The flowing medium transports the rouge particles through the entire pipeline into the dead spaces, pumps and containers where they are deposited. In pure steam systems, the rouge that forms can be strongly adherent. This deposit is dark purple (Figure 5.E-6 ).
Figure 5.E-5 Rouging in a WFI (water for injection) system (source: Henkel Beiz- und Elektropoliertechnik GmbH & Co.KG, 19306 Neustadt-Glewe, Germany)

Figure 5.E-6 Rouging in a pure steam generator (source: Ciba Spezialitätenchemie AG)

The destruction or removal of the passive film can have many different causes. If the temperature is high (e.g. above 80 °C), the ambient oxygen decreases. There is not enough oxygen to form chromium oxide. The passive film deteriorates and finally disappears (thermodynamic equilibrium). High temperatures also promote the breakdown of water into its component parts. These breakdown products (hydrogen ions H+ and hydroxide ions OH-) can also destroy the passive film because of a slight decrease in the pH value. Rouging occurs on all of the stainless steel types traditionally used in pharmaceutical facilities, e.g. 1.4435, 1.4404 and 1.4571. This is obvious from the information given above.
1 www.ispe-dach.org, Newsletter No. 1, April 2007, Rouging
How does rouging impact the water quality?
The coloured precipitates are, in chemical terms, different types of iron oxide and iron hydroxide. However, Cr and Ni-oxides or hydroxides are also present. Because the ions of these compounds are practically insoluble in water, it is not possible to detect iron ions which would mean that the water does not meet the quality requirements. A change in the specific conductivity cannot be observed either. This means that water from pipes with moderate rouging is chemically acceptable if we use the monographs in the pharmacopoeias as a reference. Even the limit value for the heavy metal content is not reached, or exceeded. The various specifications for the quality of pharmaceutical water do not contain a limit value for particles.
Figure 5.E-7 Reddish-brown deposits in a PTFE seal. The rouge particles transported in the WFI have been deposited on the seal. (Source: Henkel Beiz- und Elektropoliertechnik GmbH & Co.KG, 19306 Neustadt-Glewe, Germany)

The introduction and distribution of rouge particles from water and steam systems to production/media systems and facilities represents a particular problem. There is then a real risk that the particles will be transported into the product (the usual size of rouge particles ranges from 0.01 to 10 µm). The system must therefore be strictly controlled. If rouging occurs, suitable measures must be put in place. Rouging can often be detected on the seals (Teflon) of open systems because this is where the transported particles tend to accumulate (see Figure 5.E-7 ). As the particles are usually very small, they cannot be seen with the naked eye. Visible particles are usually larger than 50 µm.
How can rouging be addressed?
The EU GMP Guidelines state that the design, implementation and maintenance of premises and equipment must ensure that they are suitable for the intended process steps. But what happens when it is known that a material may release particles into the water and cause contamination? Rouging is a problem that should be taken into consideration during the validation or requalification of a system, and possible actions should be discussed and documented.
In practice, however, rouging is often ignored during the risk analysis and system qualification. One reason for this is that there is no great desire to include the issue in the documentation despite a proactive approach to the problem being good manufacturing practice. Despite the latest technology, rouging cannot be prevented if stainless steel is used. PVDF might be a suitable alternative. However, this material has not yet been accepted by the pharmaceutical industry, and there are technical uncertainties, with regard to hot water systems (thermal expansion), in particular.
As rouging is to be expected in hot pharmaceutical water systems, in particular, the system documentation should always cover the subject. If possible, rouging should be discussed at the planning stage. Possible measures should be documented during the risk analysis and taken into consideration during maintenance and cleaning (derouging and repassivation). In new systems, the use of electropolished surfaces can provide sufficient protection. For existing systems, accurate monitoring and documentation is recommended. Regular inspections carried out using specially installed equipment can help detect the problem at an early stage. If required, cleaning (derouging) and repassivation of the system can be carried out to restore the initial state.
The ability of rouge particles to migrate in systems that carry liquid means that rouging is not just a corrosion problem; it can also cause problems during pharmaceutical manufacture. Released particles cause an unwanted and unpredictable contamination of media and products. For this reason, a risk analysis should be carried out in each individual case to determine the impact of rouging on the quality of the manufactured products.
How can rouging be detected?
A device for measuring the rouging class is available. This inline device measures the reflectance of surfaces and the intensity of the reflected light. Polished surfaces reflect more light than surfaces that are stained with iron oxide and covered in rouging particles. The different possible states of a rouging surface are compared with the measured values, and the result is displayed (Figure 5.E-8 ).
Figure 5.E-8 Rouge monitoring in pharmaceutical water systems using the HPLG (Henkel Passive Layer Guard); left: schematic representation of the rouge display; right: practical use (source: Henkel Beiz- und Elektropoliertechnik GmbH & Co.KG. 19306 Neustadt-Glewe, Germany)

The starting point is a metal surface with a shiny finish or an electropolished surface (1). Rouge formation begins with a reduction in the passive layer (2) which leads to some initial staining of the surface - the beginning of rouging (3). When rouging increases, the alarm level is increased (4). Cleaning (derouging) and repassivation of the system should be carried out when alarm level red (5) is reached at the latest.
This relatively simple measurement makes it possible to monitor rouge formation at all times. The authorities can also see that the operator of the system proactively addresses monitoring and rouging.
Rouging can also be monitored by controlling the number of particles. This is a state-of-the-art process used in the electronics industry and is an integral part of quality control in that industry. These types of control could also be implemented in pharmaceutical water systems with very little technical effort. However, they are rarely used in the pharmaceutical industry.
Do you have any questions or suggestions? Please contact us at: redaktion@gmp-verlag.de







