
GMP Meets Radiation Protection – How Do They Fit Together?
3 min. reading time | by Andreas Nuhn
Published in LOGFILE 07/2026
The manufacture of radiopharmaceuticals requires simultaneous compliance with the EU GMP Guide and radiation protection regulations, which can lead to conflicting objectives between product safety and personal protection. Technical solutions such as negative pressure systems and lead-lined cells, as well as early consultation with the authorities, are crucial for implementation that complies with both GMP and radiation protection requirements.
In the manufacture of radiopharmaceuticals – regardless of whether they are used for diagnostics or therapy – regulations from two legal areas must be complied with. On the one hand, the products are classified as medicinal products and must therefore meet GMP requirements during manufacture, in particular Annex 1 of the EU GMP Guide on the manufacture of sterile medicinal products. Annex 3 of the EU GMP Guide, which deals with the manufacture of radiopharmaceuticals, refers to Annex 1 in many places and provides few specific requirements regarding technical aspects of product protection. Secondly, both the requirements of the Euratom Directive 2013/59 and the national requirements on radiation protection must be met.
These requirements from the various documents are contradictory in some areas. In the case of GMP requirements, patient safety and thus product safety are of paramount importance, particularly as these are parenteral products that cannot be terminally sterilised. From a radiation protection perspective, however, the technical measures must ensure that both staff in the production area and the surrounding environment are adequately protected against radioactive contamination (see Figure 1).
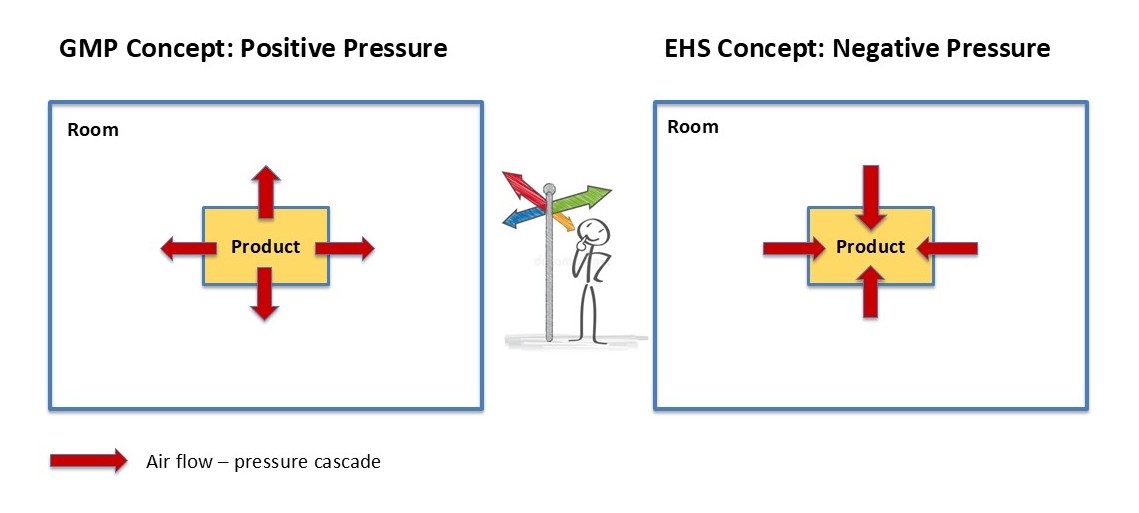
Figure 1 | Positive or negative pressure? (EHS: Environment, Health and Safety)
When working on the project, it is therefore important to involve the regulatory authorities in the technical discussions at an early stage in order to prevent costly changes to the detailed planning or, potentially, even to installations that have already been completed. Typically, radiation protection areas operate under absolute negative pressure to prevent the escape of radioactivity. For radiopharmaceuticals, lead cells are used as primary containment for both synthesis and filling. These are constructed like an isolator, but the operator can only intervene in the process during manufacture via manipulators (grippers/tongs). The processes are therefore highly automated.
To protect the environment (adjacent cleanrooms and other non-classified rooms) from radioactive radiation (contaminated air) in the event of an accident, additional negative pressure zones are installed in the non-classified area surrounding the cleanrooms (manufacturing areas). Here, coordination with the GMP supervising authority is particularly important, as the classic cascade – highest cleanliness class = highest pressure – cannot be maintained (lead cells are operated under absolute negative pressure). The cleanroom (highest pressure level) is then generally protected from external contamination by means of pressure peaks or pressure troughs. This technique is also used in the manufacture of critical biologics (from BSL 2) or highly toxic substances (from OEB 4).
Do you have any questions or suggestions? Please contact us at: redaktion@gmp-verlag.de









