Criticality classifications of deviations
An approach based on the PIC/S Guidance on Classification of GMP Deficiencies
5 min. reading time | by Felix Tobias Kern und Liwa Schneider
Published in LOGFILE 43/2020
The correct and consistent classification of deviations into the categories Minor, Major and Critical is a constant challenge for pharmaceutical manufacturers. A systematic specification is a prerequisite for the elimination of subjective components of the classifying QA function and the QPs.
This specification also prevents inhomogeneities between the individual decision-makers of the QA and between the individual QPs.
The PIC/S Guidance on Classification of GMP Deficiencies can serve as a guide in this respect.
This PIC/S Guidance is actually intended to assist GMP inspectors to achieve a risk-based and harmonised classification of GMP deficiencies. As deviations in manufacturing are largely GMP deficiencies, this guidance can also be used as a basis for a classification system of deviations.
Definitions Minor, Major and Critical according to the PIC/S Guidance
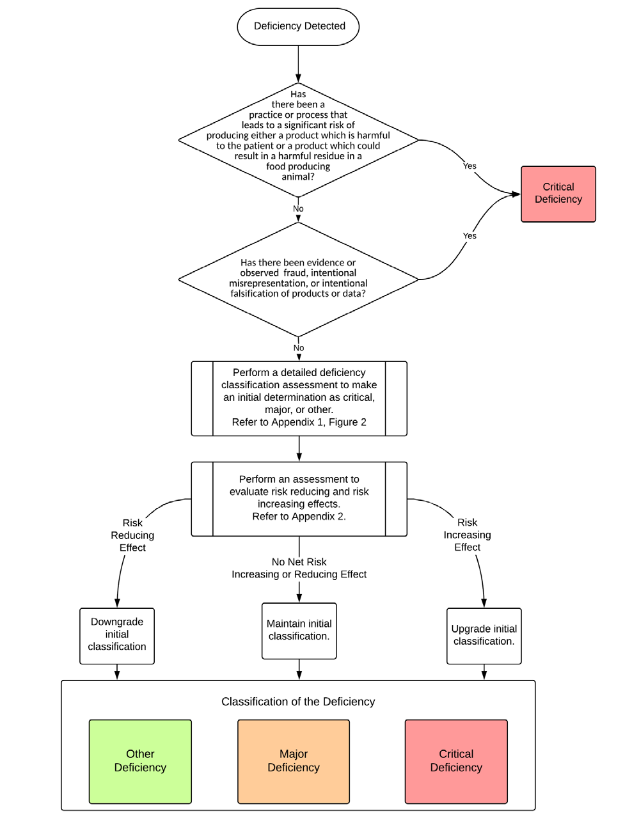
The guidance distinguishes three classes of GMP deficiencies: "Critical Deficiency", "Major Deficiency" and "Other Deficiency".
Critical
The critical deviation results in a product that endangers the patient or poses a significant risk to the patient. The time of detection, for example during production, before or after release, and the probability of detection are not considered. Therefore, if a batch is produced with a quality defect that endangers the patient, but the manufacturer notices this through representative or automatic control systems, the guidance is to classify this defect and thus the deviation as "critical".
Major
A "major deficiency" - is a deviation that is not a "critical deficiency". This means that the patient is not at risk from this deficiency/deficiency. Examples of "major" deviations are
- violation of marketing authorisation documents and specifications, manufacturing authorisation, clinical trial authorisation or pharmacopoeias
- ineffective implementation of the required GMP controls, e.g. IPC controls or other controls during production, e.g. for printing batch data on folding cartons, and the batch release test
- unreliable release procedure: This concerns both deviations concerning the system itself, e.g. release of batches without a completed investigation of a deviation that has occurred, and the persons responsible for the release
Other (Minor)
The definition of "Other Deficiency" does not correspond to the definition of the first two categories, but indicates a departure from Good Manufacturing Practice (GMP). This class can also be called "Minor Deficiency".
Risk-increasing and risk-reducing factors that can change the initial classification.
The guidance also lists additional factors that can finally increase or decrease the initial risk class.
Risk-increasing factors include the repeated occurrence of a deviation, also known as recurring. Recurring deviations are deviations that have occurred again on the same line or equipment and where CAPA measures could not prevent the deviation from occurring again. If there is a second comparable line or equipment in the production plant, this should be taken into account.
Risk-reducing factors focus on the topic of risk management. Have CAPA measures been initiated or already established on the basis of the deviation, which reduce the product, patient and/or other risks?
Summary
The PIC/S Guidance on Classification of GMP Deficiencies provides a suitable rationale for a manufacturer to establish classification rules for deviations. The Guidance contains numerous practical examples and decision trees. Furthermore, there is a list of risk-increasing and risk-reducing factors with which the initial risk assessment can be finally adjusted.
Overview of the classification process of GMP deficiencies (Source: PIC/S Guidance on Classification of GMP Deficiencies, Appendix 1, Figure 1)
Authors
Felix Tobias Kern, PhD
Pharmacist/manager of production
Merck KGaA
E-Mail: Felix.Kern@freenet.de
Liwa Schneider
Pharmacist/Deviation Manager
Merck KGaA
E-Mail: liwasschneider@gmx.de
Editorial tip:
Detailed background information and tips on the management of deviations, the exact process of handling deviations and the deviation report can be found in the GMP Compliance Adviser, in Chapter 1.E Deviations by Christian Gausepohl, PhD.
Do you have any questions or suggestions? Please contact us at: redaktion@gmp-verlag.de